Vietnam Cosmetic Regulation completely follows the ASEAN Cosmetic Directive as a member country of ASEAN. The cosmetic products are regulated by the Drug Administration of Vietnam (DAV) under the Vietnam Ministry of Health (MOH). To sell your products to the Vietnam market, you first need to go through a cosmetics registration procedure of proclamation and obtain a valid proclamation receipt number for each type of product.
Guide on Cosmetic Regulation in Vietnam
Cosmetics Definition in Vietnam
In Vietnam, Cosmetics are defined as substances or a preparation used for touch with the outer parts of a human body (skin, hair system, fingernails, toenails, lips, and external reproductive organs) or teeth and mouth mucous membrane, whose purpose is to cleanse, aromatize, change the external characteristics or form, adjust body smell, safeguard body, or maintain the human body to always be in a good condition.
If there are any differences in a cosmetic product’s shape, color, or ingredients, all variations need to be registered as separate items. For example, different shades of lipstick require separate registration.
Cosmetics Proclamation
Cosmetics proclamation in Vietnam can be done by a local business entity who is the brand owner or appointed a company by the brand owner with the function of the cosmetic business in Vietnam. In this case, for any foreign cosmetic brands, it is necessary to set your local subsidiary or appoint a local partner who has permission to trade cosmetic products in Vietnam.
Once you have set your local company or appoint a local partner, you can need to prepare and submit the following documents to proceed with your product registration in Vietnam:
- Proclamation report of cosmetics
- Company registration certificate (Copy)
- The power of attorney of the producer/owner of the product (original or notarized copy)
- Free sales certificate
- Product Information File (PIF) for every product
The PIF includes:
- Administrative documents and a summary of the product
- Material quality
- Product quality
- Safety and efficiency
Once the proclamation is completed, the Ministry of Health will issue a cosmetic product proclamation receipt for each of the products. The registration number is valid for five years since the issuance date. If you wish to extend your product sales upon its expiration, you are required to register before the expiration date.
Vietnam Cosmetics Registration Process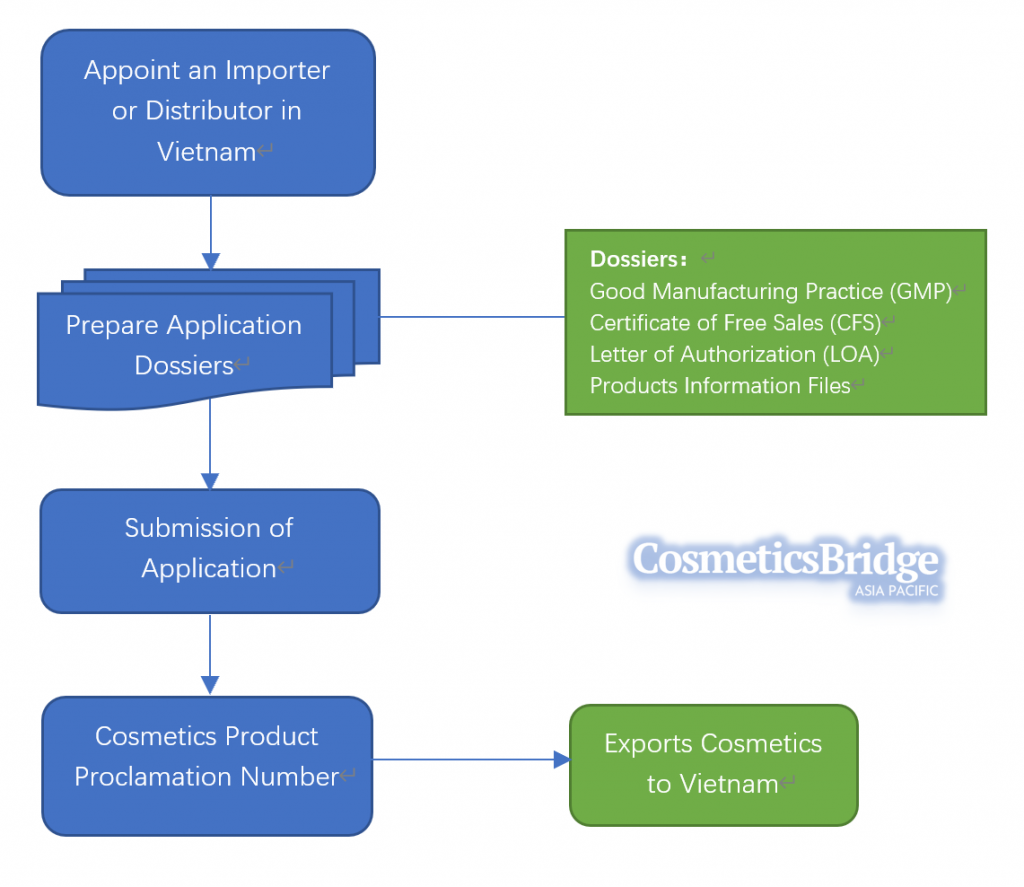
Labeling requirements
The labels must be written in English or Vietnamese and include the following information:
- Name of the product and its functions
- Usage instructions
- The full formula of ingredients
- Country of origin
- Name of the distributor
- Quantification
- Batch number
- Manufacturing or expiration date
- Warnings and precautions
Products Safety Requirement
Cosmetics are necessary to be assessed and evaluated product safety following the ASEAN safety guideline and attributes specified in Annex 06-MP. There are restrictions set out for heavy metals and microbial parameters in cosmetic products. All other requirements concerning cosmetic ingredients can be found in the latest version of the ASEAN Cosmetic regulation. The organization or individuals who are responsible for putting products on the market are organizations are also responsible for the safety, effectiveness, and quality of the product.
Access ASEAN Cosmetics Regulatory Database. ->